Background
Mosquito distributions are affected by environmental conditions which are changing constantly with the onset of anthropogenic climate change (Gorris et al., 2021). These environmental changes, such as increased temperatures, influence the distribution of vector-borne pathogens through changes in the geographic distribution, transmission dynamics, and the re-emergence of vector-borne diseases in previously unaffected areas (Rocklöv & Dubrow, 2020). Defining and tracking the geographical distributions of disease vectors is critical to employing provisions to mitigate the spread of these vector-borne diseases (Gorris et al., 2021).
Mosquitoes are one of many adaptable and widespread organisms that transmit vector-borne pathogens worldwide (Anosike et al., 2007). Like other arthropods, mosquitoes emerge and reproduce when seasonal temperatures increase, allowing for proper development and reproduction to occur (Drakou et al., 2020). In response to various environmental changes such as increased temperatures and alterations to the relative humidity and precipitation, mosquito populations have been shown to alter their development, reproduction, and vector abundance. These alterations affect the geographic spread, activity, and overall mortality rates of the mosquito populations (Roiz et al., 2014).
The Culex pipiens complex is a species complex – a group of closely related species that can be extremely difficult to be distinguished morphologically but can be identified at the molecular level (Sousa-Paula et al., 2021). The species within the Culex pipiens complex includes six contemporarily recognized members, Cx. quinquefasciatus, Cx. pipiens f. pipiens, Cx. pipiens f. molestus, Cx. pipiens pallens, Cx. australicus and Cx. globocoxitus (Aardema et al., 2020). The species of the Cx. pipiens complex are morphologically identical among females, differing only in their behavioral and physiological characteristics (Shaikevich et al., 2016). The members of the complex can be identified by the male phallosome, but this trait cannot identify anautogenous females (Joyce et al., 2018). The lack of distinguishable morphological differences in the female mosquitoes makes surveillance efforts difficult (Farajollahi et al., 2011).
The Culex pipiens complex are important, widespread vectors (Shaikevich et al., 2016). The members of this complex are known as competent enzootic vectors for a variety of arboviruses including St. Louis encephalitis, West Nile virus, Western Equine Encephalitis, and Malaria (LaBeaud et al., 2011; Simon et al., 2022; Manuela, et al., 2021). This vectorial competency arises from the diversity within this complex, especially pertaining to the feeding habits among the various members of this complex, resulting in discontinuities in disease transmission dynamics (Farajollahi et al., 2011). The members of the Cx. pipiens complex possess putatively different transmission rates, with Cx. quinquefasciatus possessing a higher vectorial capacity for West Nile virus than others in the complex (Ciota et al., 2013). This difference in vectorial capacity suggests a need to redefine the distribution of the Cx. pipiens complex to monitor the transmission of West Nile virus and other arboviruses carried by the Cx. pipiens complex. Accurate identification of this complex is critical for surveillance of West Nile virus because of the recognized differences in vectorial capacity within the complex (Farajollahi et al., 2011; Smith & Fonseca, 2004).
Various morphological studies have established the hybrid zone of the Culex pipiens complex in North America (Joyce et al., 2018; Kothera et al., 2009). In 2004, the population dynamics of the Cx. pipiens complex was established in California using molecular analysis, with Cx. pipiens pipiens occurring North of 39° latitude and Cx. quinquefasciatus occurring South of 36° latitude. Among these latitudes, the two species overlap and a hybrid species can occur (Joyce et al., 2018). The Cx. quinquefasciatus prefer warmer temperatures and persist later into transmission seasons (Farajollahi et al., 2011). The population dynamics of the Cx. pipiens complex established in California was used as a baseline for this study.
Understanding the distribution of the Cx. pipiens complex is not only important for vector transmission dynamics, but also in understanding the effects anthropogenic climate change will have on this complex within the intermountain west. As Cx. quinquefasciatus favors warmer temperatures, it is possible that their distribution will shift northward as global temperatures increase, but in order to observe this effect, reliable population studies must first establish a baseline. This study is the first to determine the molecular distribution of the Cx. pipiens complex in Utah. Additionally, this study can serve as a model to promote surveillance of the various arboviruses transmitted by this complex for vector management programs along with providing information to make comprehensive decisions to support communities throughout the state of Utah.
Methods
Collection of Specimens
During June, July, and August of 2021, various sampling sites were chosen in multiple counties throughout the state of Utah, including Washington, Box Elder, Tooele, Utah, and Salt Lake Counties. Specimens were collected using CO2 light traps. Geographic origins of the specimens, trapping date, and the number of specimens and the identified species are listed in Table 1.
Molecular Analysis
This study adapted the PCR-based ACE Rapid Assay that amplifies polymorphisms in the second intron of the nuclear genes of the acetylcholinesterase-2 (ace-2) locus which was found to be species-specific, distinguishing among Cx. pipiens, Cx. quinquefasciatus and their hybrids (Smith & Fonseca, 2004).
Genomic DNA was extracted from individual Cx. pipiens and Cx. quinquefasciatus female mosquitoes were homogenized in a 5% Chelex 100 solution using a BeadGenie (Scientific Industries). Following DNA extraction, PCR was adapted and carried out using parameters from the ACE Rapid Assay, using the following primers: ACEpip, ACEquin, and B1246s (Smith & Fonseca, 2004). The reactions were run in a SimpliAmp Thermal Cycler (Applied Biosystems) at the following parameters: 94°C for 2 minutes, 35 cycles at 94°C for 30 seconds, 56°C for 30 seconds, 72°C for 30 seconds, followed by a final extension temperature of 72°C for 2 minutes. Following PCR, samples were run in a 2% agarose gel and visualized on a BioRad GelDoc Go Gel Imaging System.
Results
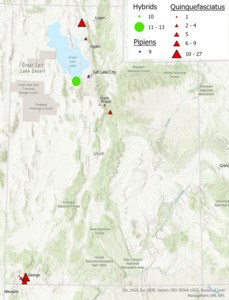
Contemporary presence data was gathered for the Cx. pipiens mosquito species complex in various counties in Utah. Molecular identification of the Cx. pipiens pipiens, Cx. quinquefasciatus, and their hybrids was carried out using the ACE Rapid Assay. The geographical distribution of the Cx. quinquefasciatus, Cx. pipiens pipiens, and their hybrids in various counties throughout the state of Utah is illustrated in Figure 1.
Our results indicate that the majority of the Culex complex mosquitoes in Utah are Cx. quinquefasciatus. Hybrids and Cx. pipiens pipiens were identified in Salt Lake county. In the southern region of the state, all specimens identified were Cx. quinquefasciatus.
Conclusion
These findings suggest that a majority of the state of Utah has a higher transmission rate of West Nile virus due to the presence of the Cx. quinquefasciatus which has a higher vectorial capacity for West Nile virus than other members of the complex.
The observation that Cx. quinquefasciatus were the only species identified, ranging from Washington county in the south, and Utah county in the northern part of the state establishes an important baseline. Previous distribution maps predicted that the hybrid zone would fall within the southern region of the state, but this study reveals that the hybrid zone is much further north than expected. This could be due to lack of molecular studies of this complex leading to inaccurate predictions, but could also be the result of anthropogenic climate change. Further studies using museum specimens would further illuminate the cause.
Defining the contemporary geographical distributions of the Cx. pipiens complex in Utah is critical to tracking the spread of the various arboviruses transmitted by this vector, including West Nile virus. The distribution of the Cx. pipiens complex mosquitoes will continue to shift in response to anthropogenic climate change, shifting geographical distributions of disease vectors, and exposing new populations to mosquito-borne diseases. Understanding the population dynamics and the spatial distribution of the Cx. pipiens complex can assist in providing disease mitigation strategies from vector management programs, and decision support for communities throughout the state of Utah.